This gives it Ax2E2 geometry and so it is V-Shaped/Non-linear/Bent. The sulfur, on the other hand, has two sigma bonds (single bonds on either side) AND two lone pairs. The carbon atom is only bonded to two other atoms and has no lone pairs this gives it AX2 geometry, and so there is a linear arrangement around the carbon atom. This means is does not need any leftover p orbitals (which would need to stay unhybridized) and so sulfur is “sp3” hybridized here. C) atomic orbitals of the bonding atoms must overlap for a bond to form.
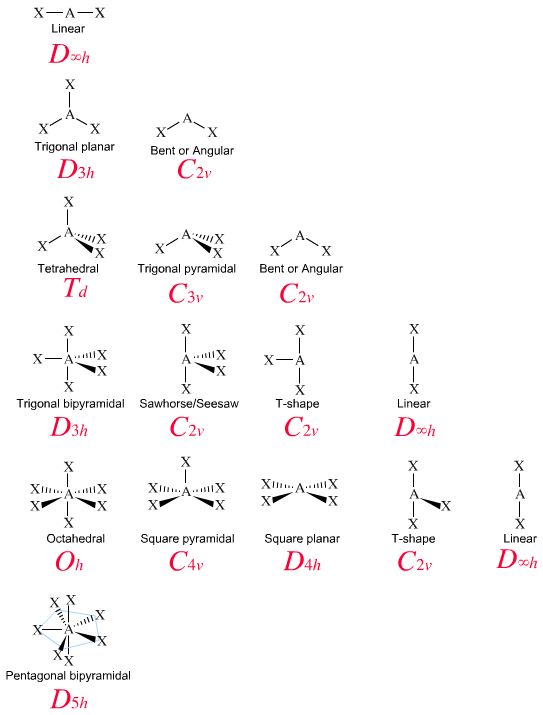
B) regions of electron density in the valence shell of an atom will arrange themselves so as to maximize overlap. A) regions of electron density on an atom will organize themselves so as to maximize s-character. It has two single bonds (both sigma) and two lone pairs. 1) The basis of the VSEPR model of molecular bonding is. The sulfur atom has NO double or triple bonds. The sigma bond (first bond between the two is sigma) and the lone pair are where the “hybridized sp orbitals” are used here.

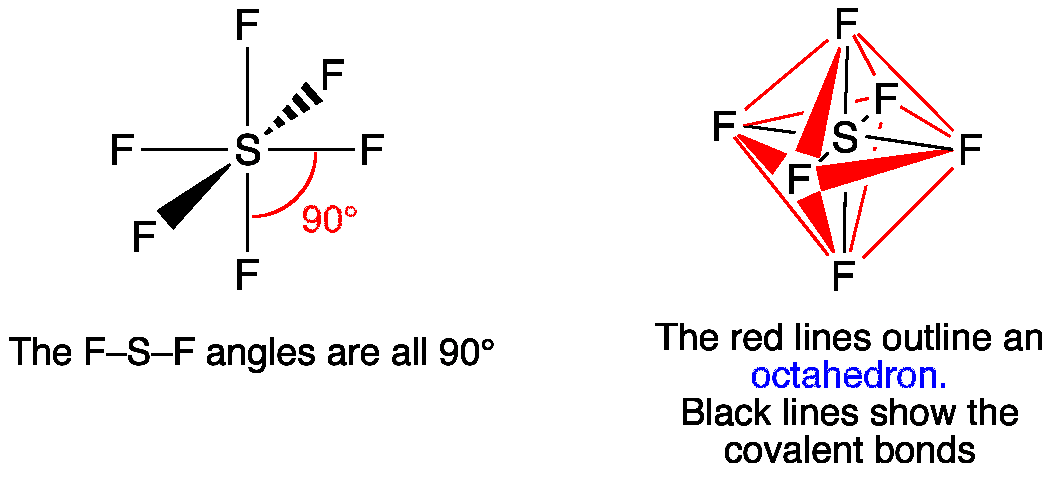
N is also triple bonded, so it is “sp” hybridized as well. HSCN has a single bond between H and S, a single bond between S and C, and a triple bond between C and N.Ĭarbon is triple-bonded to nitrogen this requires two pi bonds and that means it requires two leftover p orbitals after hybridization so the C is “sp” hybridized. What is the molecular geometry of scl2 The molecular geometry of SCl 2 is AX 2 E 2 or bent/angular/V-shaped. This also gives sulfur a seventh electron, and it gets its eighth from hydrogen. So according to VSEPR theory, the electron geometry of SF2 S F 2 molecule will be tetrahedral which includes all the valence electrons of the central atom as shown in the figure below. Meanwhile, carbon needs one more electron and sulfur can provide it. Ammonia also has four electron pairs and the coordination geometry of nitrogen is based upon a tetrahedral arrangement. Carbon and nitrogen share three electrons each (with each other) and a triple bond forms between them. Nitrogen brings five valence electrons, and so it needs three to completed its octet.
All four of these atoms are non-metals, the bonds between them will be covalent, and it will make a molecular compound.Ĭarbon brings four valence electrons with it, and so needs four more to complete its valence shell. HSCN is the corresponding acid of SCN-, which is the thiocyanide ion.
